
Newsroom
IRF3 Found to Play Novel Role in Regulating Hypoxia Signaling
Upon viral infection, interferon regulatory factor 3 (IRF3) undergoes phosphorylation and activation. It then translocates to the nucleus, where it dimerizes and functions as a key transcription factor that initiates the innate antiviral response. In the resting state, IRF3 is located in the cytosol and is inactivated. However, its function in the cytoplasm during non-infection states remains unclear.
A research group led by Prof. XIAO Wuhan from the Institute of Hydrobiology (IHB) of the Chinese Academy of Sciences identified that IRF3 is a negative regulator of hypoxia signaling by retaining hypoxia-inducible factor α (HIF-α) in the cytoplasm. The study has been published in Cell Reports.
In this study, the researchers discovered that IRF3 negatively regulates the hypoxia signaling pathway. The suppressive role of IRF3 on hypoxia signaling depends on its localization in the cytosol. Knockout of IRF3 enhances hypoxia response gene expression and potentiates cellular hypoxia adaptation.
Mechanistically, the researchers revealed that under hypoxic conditions, cytoplasmic IRF3 interacts with HIF-1α and HIF-2α, two master regulators of hypoxia signaling. This interaction prevents HIF-1/2α from entering the nucleus and exerting its transcription factor function, thereby attenuating hypoxia signaling.
This study reveals the role of IRF3 in non-infection states by modulating the hypoxia signaling pathway and provides insight into novel crosstalk between two important ancient stress signaling pathways, both the innate immunity pathway and the hypoxia pathway.
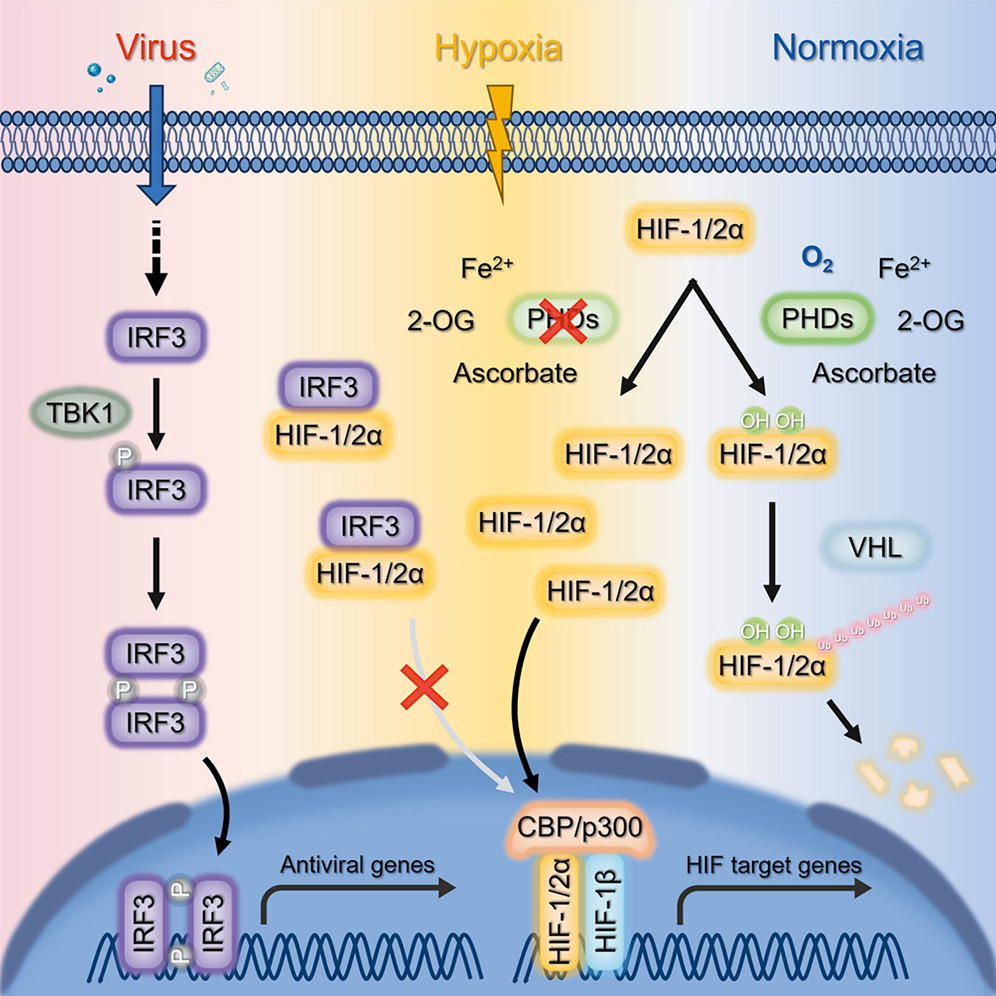
A working model for the role of IRF3 under hypoxic conditions or upon viral infection (Image by IHB)
(Editor: MA Yun)
Follow us on Facebook