
Newsroom
Engineering iTreg-mediated Immune Tolerance in Zebrafish Overcomes Allograft Rejection
The generation of genetically modified lines in model and economically species typically requires months or even years. Developing strategies to accelerate this process is therefore a critical need in the field. Transplantation of allogeneic gonadal primordia into near-mature recipients offers a promising approach for rapid gamete production. However, a major challenge is overcoming immune rejection of donor tissues.
A key question is whether immune tolerance can be induced in hosts by leveraging the central role of Foxp3-mediated regulatory T cells (Tregs) in maintaining immune tolerance.
Recently, a research group led by Prof. Sun Yonghua from the Institute of Hydrobiology (IHB) of the Chinese Academy of Sciences established an inducible Treg (iTreg)-mediated immune-tolerant host that overcomes the immune rejection barrier in subcutaneous transplantation of allogeneic gonadal primordia. This study was published in Journal of Genetics and Genomics.
In this study, the team first generated an immune-tolerant zebrafish line, Tg(CMV:foxp3a), by overexpressing the core Treg transcription factor foxp3a. Unlike immunodeficient models, this line exhibits normal growth and fertility under standard conditions. Multi-omics analyses revealed that foxp3a overexpression reshapes the immune landscape by reducing effector T and B cells involved in rejection, while increasing resting T cells that can rapidly differentiate into functional iTregs, thereby enhancing immune tolerance.
To accelerate donor gonad development, allogeneic gonadal primordia were isolated and transplanted into the dorsal subcutaneous region of 2-month-old wild-type hosts depleted of endogenous germ cells. In wild-type fish, all grafts were completely rejected within 14 days, and ectopic gonads failed to develop. In contrast, when Tg(CMV:foxp3a) fish were used as hosts, approximately one-quarter supported successful engraftment and expansion of donor tissue, forming enlarged ectopic gonadal masses, demonstrating effective iTreg-mediated immune tolerance.
Further analysis showed that donor-derived germ cells within these grafts underwent ultra-rapid spermatogenesis in 2-month-old recipients, producing donor-derived transgenic or gene-edited F1 offspring within approximately one month.
In summary, foxp3a overexpression reprograms the immune microenvironment of hosts, overcoming allograft rejection and enabling ultra-fast maturation of donor-derived gametes. This study establishes an immunoregulatory foundation for large-scale applications of allogeneic surrogate reproduction, and provides a new strategy for rapid generation of genetically modified lines in both model and economically important species.
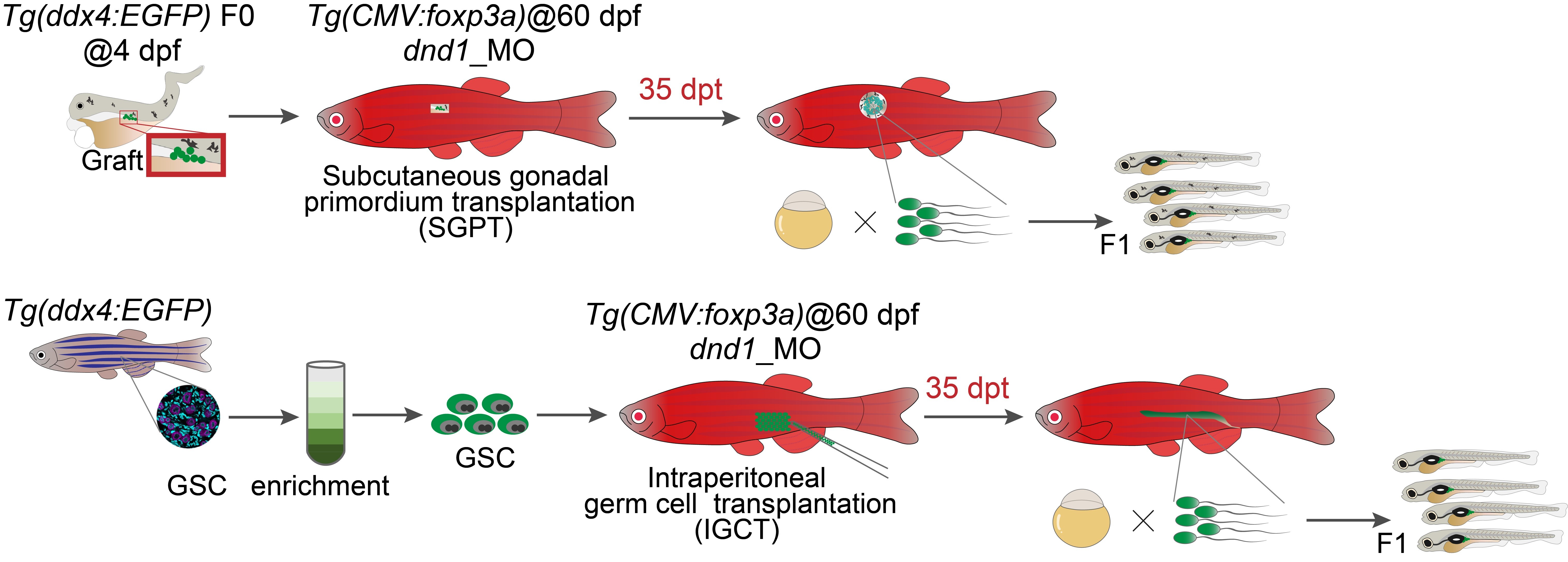
iTreg-mediated immune tolerant zebrafish enable ultra-rapid strain generation via SPGT and IGCT (Image by IHB)
(Editor: MA Yun)