Highlights
Oxidative Stress Facilitates Infection of Unicellular Alga Haematococcus pluvialis by Fungus Paraphysoderma sedebokerense
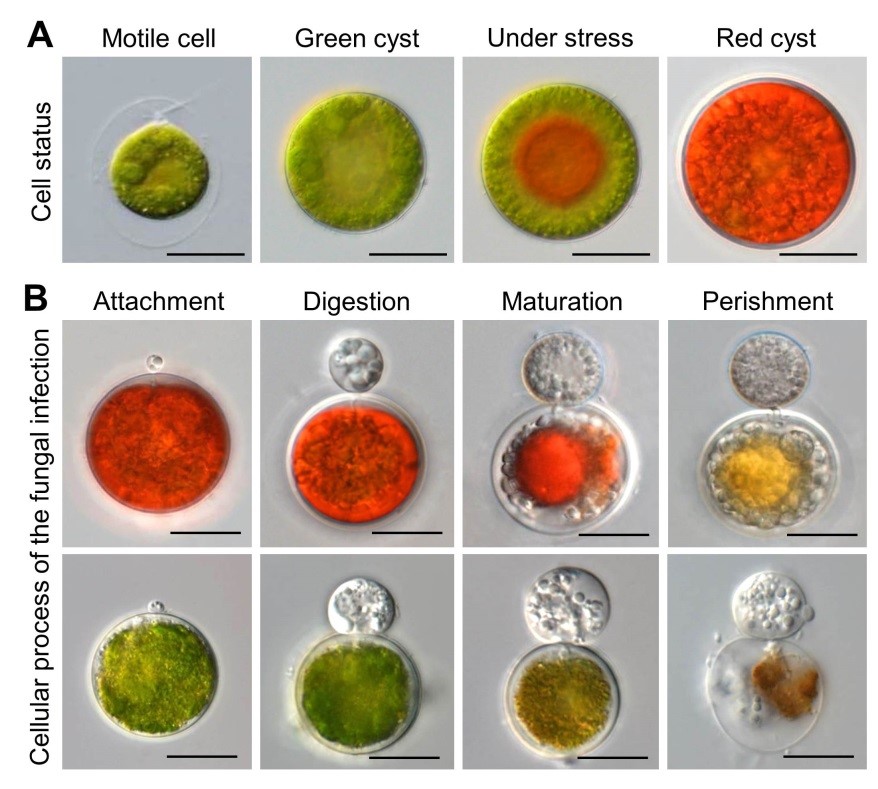
The green unicellular microalga Haematococcus pluvialis is considered as the most sustainable feedstock for the commercial production of astaxanthin. However, the development of H. pluvialis mass culture industry has been retarded by the contaminations of various microbial pathogens, among which is a parasitic fungus Paraphysoderma sedebokerense that shows highly species-specific infection on H. pluvialis cells and causes the most destructions and economic losses to the mass culture of H. pluvialis all over the world.
The underlying infection mechanisms of the fungal pathogen P. sedebokerense on H. pluvialis hosts remain largely unknown, making it rather challenging to develop effective measures to mitigate the pathogen threatening.
Recently, a research group lead by Professor HAN Danxiang from the Institute of Hydrobiology (IHB) of the Chinese Academy of Sciences found that secondary metabolites mediated oxidative stress facilitated the infection processes by causing decreased resistance of H. pluvialis to the fungal parasitism. Based on the discovery, a biosafe and environmentally-friendly chemical was screened to block the oxidative stress and showed effective infection inhibitory effect in the scale-up culture of H. pluvialis. The research was published in Biotechnology for Biofuels and bioproducts.
In this study, the researchers first observed that the supernatants of the infected cell culture significantly enhanced the infection process, and directly degraded the pigments and carbohydrates of the algal hosts. The effectors could not be removed by filtration with 3K Da membrane, suggesting they were small molecular metabolites.
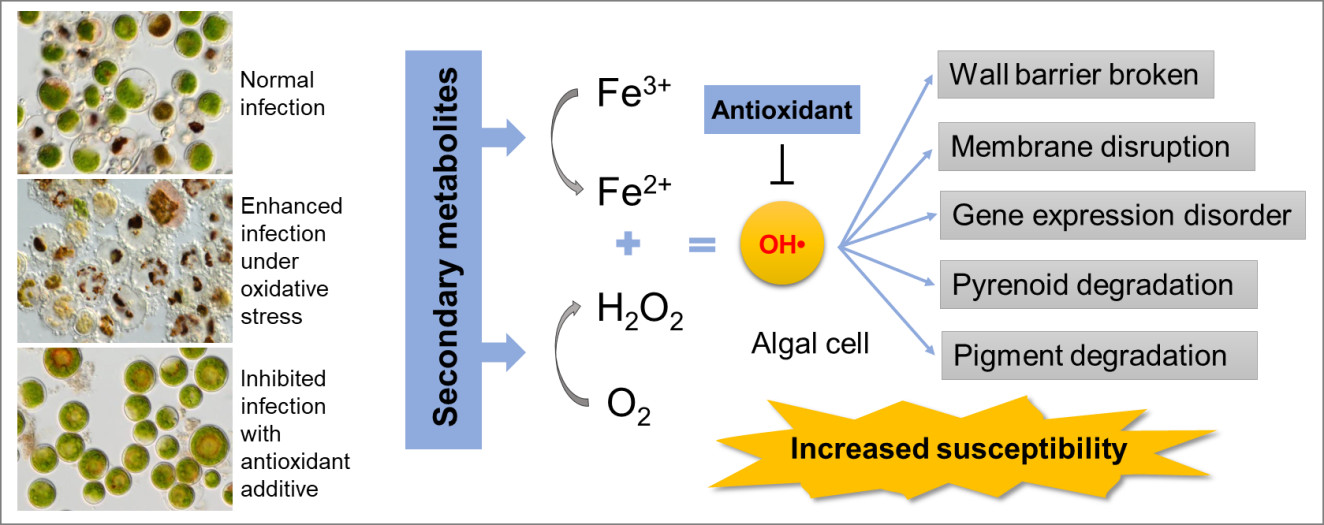
In further experiments, transcriptomic analysis indicated that the supernatant post infection (SPI) triggered oxidative stresses on the algal cells. The researchers then detected high oxidative activity of the SPI both in vitro and in vivo and proved the SPI could produce hydroxyl radical. Based on these results, they hypothesized that the damage-associated metabolites produced reactive oxygen species by triggering Fenton-like reactions.
The researchers further conducted time-course metabolomics analysis and retrieved 10 metabolites with suspected structures from the metabolites. They used the commercial products to investigate the activity of these candidates, and found that hordenine and 3-hydroxyanthranilic acid (3-HAA) significantly reduced the cellular contents of carbohydrates and pigments, and both of these metabolites stimulated the infection process.
In addition, 3-HAA showed strong activity in reducing Fe3+ to Fe2+. 3-HAA and hordenine induced the algal cells to produce H2O2, indicating that 3-HAA and hordenine were responsible for the Fenton reaction-induced oxidative stress in the SPI, as Fe2+ and H2O2 were two important participants in the Fenton reaction.
Based on these findings, the researchers selected an antioxidant butylated hydroxyanisole (BHA) to inhibit the fungal infection by eliminating the oxidative stress. The infection was completely abolished both at experimental level and in the 100 L open raceway ponds, further demonstrating that oxidative burst was a strategy for the pathogens to infest.
This study provided a framework that could be leveraged to dissect the functions of secondary metabolites in the interaction between the unicellular algal and true fungi. It would serve as a guide for developing rational crop protection measures to improve the sustainability of algal mass cultivation by utilization of antioxidants.