Highlights
Researchers Reveal Role of Methyltransferase SMYD3 in Hypoxia Tolerance
Oxygen profoundly affects physiology of aerobic organisms through multiple mechanisms. Inadequate oxygen availability (hypoxia) can lead to cellular dysfunction and even cell death. In the process of hypoxia adaptation, the hypoxia signaling pathway mediated by hypoxia-inducible factor (HIF) plays a pivotal role. The factors affecting hypoxia signaling pathway mainly impact on HIFα protein stability. To identify novel regulators of HIFα protein stability will sheds new lights on the knowledge in hypoxia adaptation or tolerance
A research group led by Prof. XIAO Wuhan from the Institute of Hydrobiology (IHB) of the Chinese Academy of Sciences identified that methyltransferase SET and MYND domain-containing protein 3 (SMYD3) impairs hypoxia tolerance by augmenting hypoxia signaling independent of its enzymatic activity. The study was published in the Journal of Biological Chemistry.
In this study, the researchers found that, in a range of methyltransferases, only SMYD3 is significantly suppressed under hypoxia, indicating that SMYD3 might play vital roles in hypoxia response. By overexpression or knockout SMYD3 in cells, they verified that SMYD3 augments hypoxia signaling, which is independent of HIF1α hydroxylation and pVHL intactness.
Through co-immunoprecipitation and immunofluorescence analysis, the researchers confirmed that SMYD3 binds to and stabilizes HIF1α, leading to an increase of nuclear HIF1α and enhancement of HIF1α-mediated target gene expression, independently of its methyltransferase activity.
In addition, the researchers revealed the biological consequences of the transcriptional activity enhancement of HIF1α by SMYD3, and found that SMYD3 induces reactive oxygen species (ROS) accumulation and enhances hypoxia-induced apoptosis.
Taking advantage of zebrafish model, Xiao’s team further analyzed the in vivo function of SMYD3 in hypoxia response. They knocked out smyd3 in zebrafish via CRISPR/Cas9 technology and revealed that disruption of smyd3 in zebrafish facilitates hypoxia tolerance.
This study not only reveals novel function of SMYD3, but also uncovers new regulatory models of hypoxia tolerance by SMYD3, which will help to select candidate targets for breeding fish strains with hypoxia tolerance by CRISPR/cas9 technique in aquaculture industry.
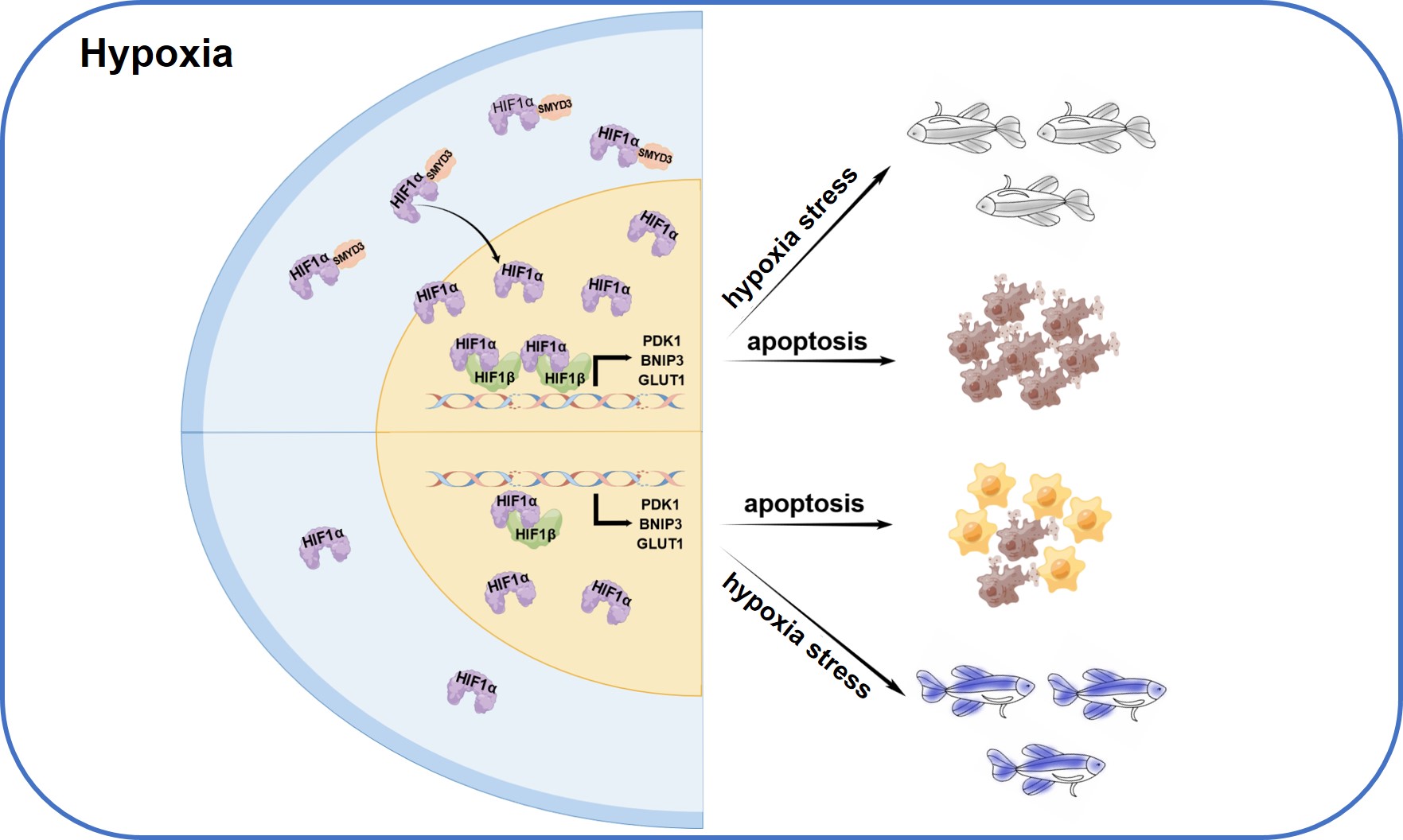
The working model for SMYD3 in hypoxia stress response (Image by IHB)
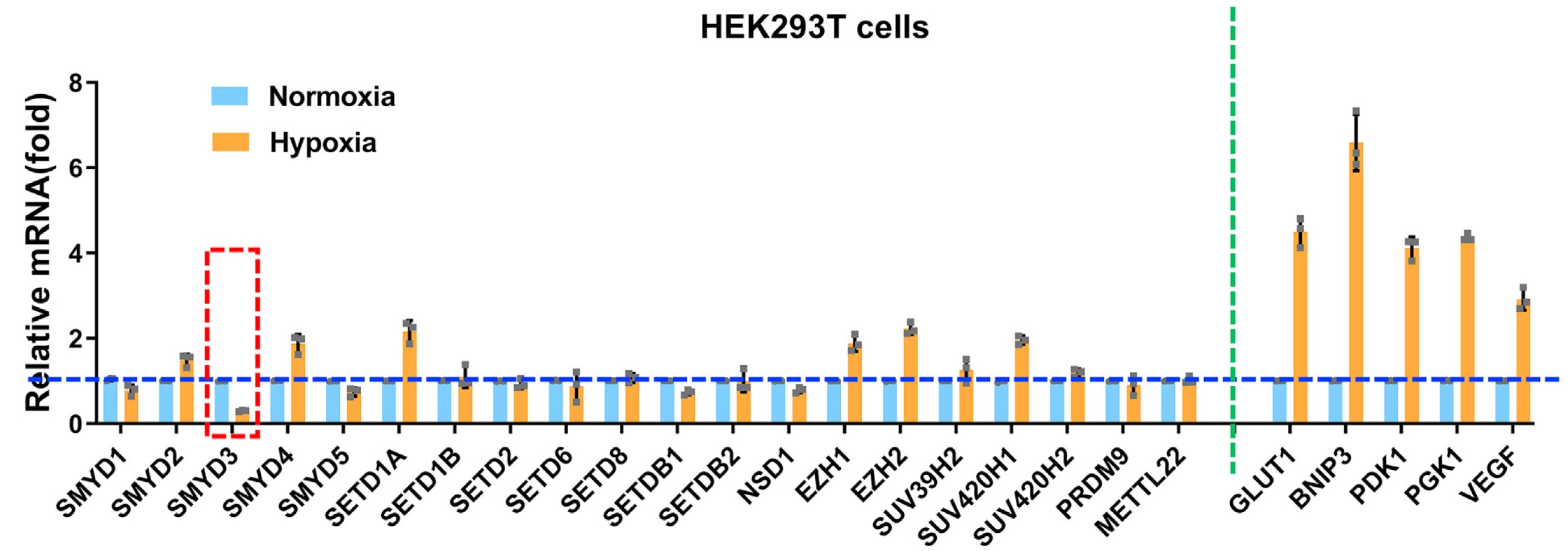
Expression of SMYD3 was significantly suppressed under hypoxia (Image by IHB)

Disruption of smyd3 in zebrafish facilitates hypoxia tolerance (Image by IHB)
(Editor: MA Yun)