Highlights
Efficient Approach to Synthesize Sterile Allopolyploids in Polyploid Carassius Complex
Polyploidy is generally thought to be a main driving force of speciation, as the polyploids possess significant advantages in genome buffering, heterosis fixing, and plasticity evolving. From the perspective of agriculture and aquaculture, polyploidy can be considered a key factor in crop and fish domestication as polyploids often have some economically favorable traits, such as increased yield, improved quality, and enhanced adaptability. Many major crops and farmed fishes are proposed to have arisen from one or two polyploidy events.
More importantly, polyploidy may lead to reproduction mode transition by favoring the onset of unisexual reproduction. More than 60% of unisexual vertebrate species or biotypes are polyploids, and some of them coexist sympatrically with a closely related bisexual diploid species, forming sexual diploid-unisexual polyploid complexes. Interestingly, some complexes display a variety of reproduction strategies.
Recently, a research team led by Prof. GUI Jianfang from the Institute of Hydrobiology (IHB) of the Chinese Academy of Sciences discovered a unique reproduction mode in the polyploid Carassius complex and developed an efficient approach to synthesize sterile allopolyploids. The study was published in Science Bulletin.
The polyploid Carassius complex is naturally composed of sexual amphidiploid (A2n) C. auratus and gynogenetic amphitriploid (A3n) C. gibelio. By integrating the genomes of the gynogenetic C. gibelio and sexual C. auratus, the researchers generated novel amphitriploids (NA3n) and found gynogenesis was recovered in most NA3n females (NA3n♀I).
In a few NA3n females (NA3n♀II), the researchers discovered a unique reproduction mode, termed ameio-fusiongenesis, which combines the abilities of both ameiotic oogenesis and sperm-egg fusion. These females inherited ameiotic oogenesis to produce unreduced eggs from gynogenetic C. gibelio and sperm-egg fusion from sexual C. auratus. Discovery of the unique reproduction mode provided an opportunity to efficiently synthesize some novel allopolyploids with higher ploidy.
The researchers subsequently utilized this unique reproduction mode to generate a group of synthetic alloheptaploids by crossing NA3n♀II with Megalobrama amblycephala. The synthetic alloheptaploids contained all chromosomes of maternal NA3n♀II and a chromosomal set of paternal M. amblycephala. Intergenomic chromosome translocation between NA3n♀II and M. amblycephala were also observed in a few somatic cells.
By further exploring the cytological mechanism underlying infertility of synthetic alloheptaploids, the researchers examined the assembly of synaptonemal complex, dynamic changes of DNA double-strand break (DSB) formation and repair in primary oocytes and spermatocytes during meiotic prophase I. Primary oocytes of the alloheptaploid underwent severe apoptosis owing to incomplete DSB repair at prophase I. Although spermatocytes displayed similar chromosome behavior at prophase I, they underwent apoptosis due to chromosome separation failure at metaphase I.
Unfortunately, massive production of the sterile alloheptaploids requires large amounts of NA3n♀II. The researchers thus established a sustainable clone for the large-scale production of NA3n♀II with the ameio-fusiongenesis mode and developed an efficient approach to synthesize diverse allopolyploids containing genomes of different cyprinid species.
This study uncovers a unique reproduction mode in the polyploid Carassius complex and provides new insight into the association between polyploidy and reproduction transition.
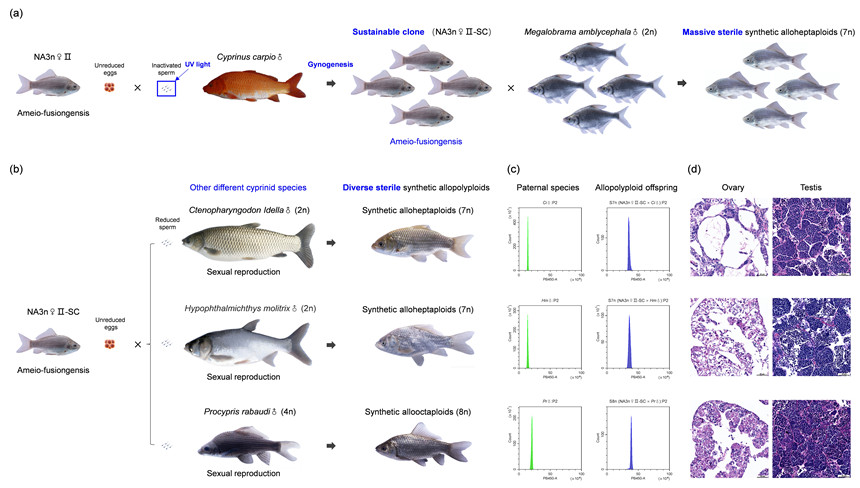
Sustainable clone establishment and diverse allopolyploid synthesis (Image by IHB)
(Editor: MA Yun)